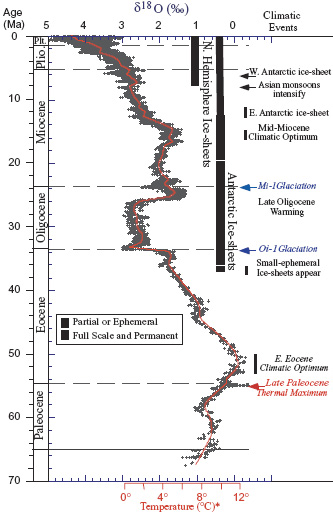
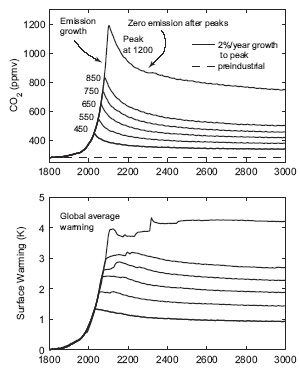
The equivalent of ~25% of current anthropegenic CO2 additions to the atmospheric reservoir is buffered by the oceans:
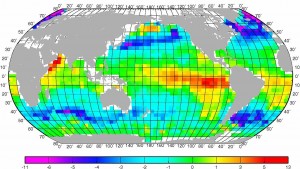
Dissolved CO2 exists as different species: CO2(aq), HCO3-(aq) and CO3=(aq). Ocean pH is the dominant control on this speciation, itself a function of pCO2:

CO2 speciation greatly affects CO2 fluxing across the ocean-atmosphere boundary, as only non-ionized species (that is, CO2(aq)) are free to exchange with the atmosphere. Anthropogenic increases in ocean acidity can be expected to greatly lower the CO2 buffering capacity of our oceans.